The accuracy of a soil test is limited, in part, by the quality of the tested sample. For this reason, strong emphasis is placed on ensuring representative samples are collected in the field. However, these samples must also be handled properly after they have been collected.
Soils are home to a diverse population of microorganisms, many of which help decompose crop residue and cycle nutrients in soils. This nutrient cycling is crucial for crop production, but can skew soil test results if it continues in soil samples after they have been collected.
Microorganisms drive the soil nitrogen cycle
The nitrogen (N) cycle in soils is particularly complex and is strongly influenced by microbial activity and, therefore, temperature and soil moisture conditions. Bacteria and fungi consume organic material and use carbon as an energy source. During this process, N contained in the organic matter undergoes several transformations, ultimately converting it to ammonia. This conversion from organic-N to inorganic-N (NH4+, ammonium) is called “mineralization.” Plants can then take up the ammonium (NH4+), or converted to nitrate (NO3-) by certain bacteria through a process known as “nitrification”.
The microbial activity requires moisture and heat, and the processes described above happen more quickly in warm, wet soils than in cold, dry soils. Microbial activity does not stop just because a sample has been collected and put in a bag. This activity continues as long as the environmental conditions are favorable. As a result, soil tests for plant-available N have the potential to change substantially if samples are not handled properly. This is an important consideration for growers because these soil test results are used to determine the profile-N credit and, ultimately, adjust N fertilizer recommendations.
Research study on soil sample storage
A recent study at the K-State Soil Testing Lab illustrates what can happen if sample submission is delayed. For this study, soil was collected from the Agronomy North Farm (Manhattan, KS) and thoroughly mixed/sieved to homogenize the material. This soil was then placed into sample bags, which were randomly assigned to different combinations of storage temperature and duration. One set of samples was kept in a refrigerator while the other set was kept in a cargo box in a truck bed. To monitor changes in soil test levels over time, three sample bags were removed from the refrigerator and truck box every two days (48 hours) and tested in the lab.
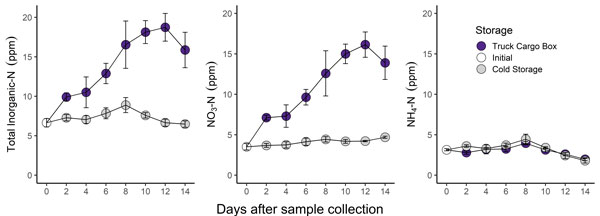
Figure 1. Change in soil test nitrogen parameters over a 14-day storage period. Samples stored in an unrefrigerated cargo box are indicated by purple points. Samples stored in a refrigerator (38F) are indicated by grey points. Graphs by Bryan Rutter, K-State Research and Extension.
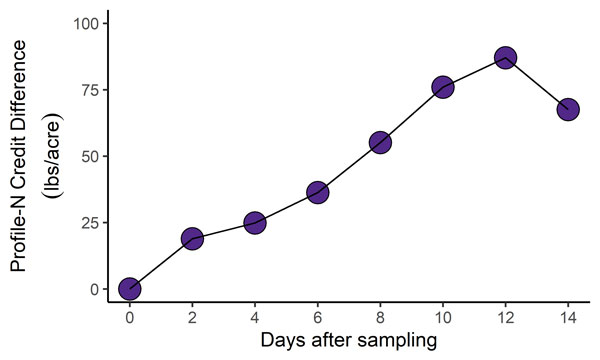
Figure 2. Difference in the soil test nitrogen credits between refrigerated and unrefrigerated samples over a 14-day storage period. Profile-N credits assume a 24-inch profile soil sample depth, and are calculated as: N ppm x 0.3 x 24 inches. Graph by Bryan Rutter, K-State Research and Extension.
Take home points from the K-State Soil Testing Lab study:
- Mineralization and nitrification led to more than a 3x increase in soil test nitrate in the undried and unrefrigerated “Truck Cargo Box” samples (purple points in Figure 1).
- Soil test nitrogen did not change substantially in refrigerated samples.
- Profile-N credits calculated from soil test N results were nearly 100 lbs of N/acre higher for the unrefrigerated samples (Figure 2).
- Improper handling and storage of soil samples can dramatically reduce soil test accuracy and may lead to under or overfertilizing crops.
K-State Soil Testing Lab Recommendations
- Submit soil samples to the lab as soon as possible, ideally on the same day they were collected.
- If same-day submission is not possible, samples should be air-dried or placed in a refrigerator set at 40 degrees F or less.
Please see the accompanying article “The challenge of collecting a representative soil sample” for guidance on field soil sampling practices.
For detailed instructions on submitting soil samples to the K-State Soil Testing Lab, please see the accompanying article “Fall soil sampling: Sample collection and submission to K-State Soil Testing Lab”.
For detailed information on how N credits are calculated please see the MF-2586 fact sheet: “Soil Test Interpretations and Fertilizers Recommendations”.
Bryan Rutter, PhD student and Soil Testing Lab Manager
rutter@ksu.edu
Dorivar Ruiz Diaz, Soil Fertility Specialist
ruizdiaz@ksu.edu
Tags: soil sampling soil testing nitrate